cc-tdi
DEVELOPMENT PIPELINE
The Children’s Cancer Therapy Development Institute is a 501c3 nonprofit R&D lab supported by federal grants, foundation grants, families and grassroots philanthropists (these philanthropists are sometimes literally 11 year-olds hosting bake sale stands).

It takes a village to address the unmet clinical need of uncommon conditions such as pediatric cancers, but we are steadfast in our mission to make all childhood cancers universally survivable by bringing basic science discoveries and science-justified, hope-filled new treatments to clinical trials. Your support makes this mission happen.
Below is the cc-TDI Pipeline.
What happens after the R&D? Artisan Biopharma is a for-profit* public benefit corporation owned entirely by cc-TDI. Artisan is the connection for moving breakthrough research and development at cc-TDI into transformative clinical trials. The Artisan mission is to provide research-driven interventions for the treatment and prevention of cancer in children.
Want to get involved? Email Charles at charles@cc-tdi.org.
* The FDA requires that clinical trial approval applications be filed by a for-profit entity.
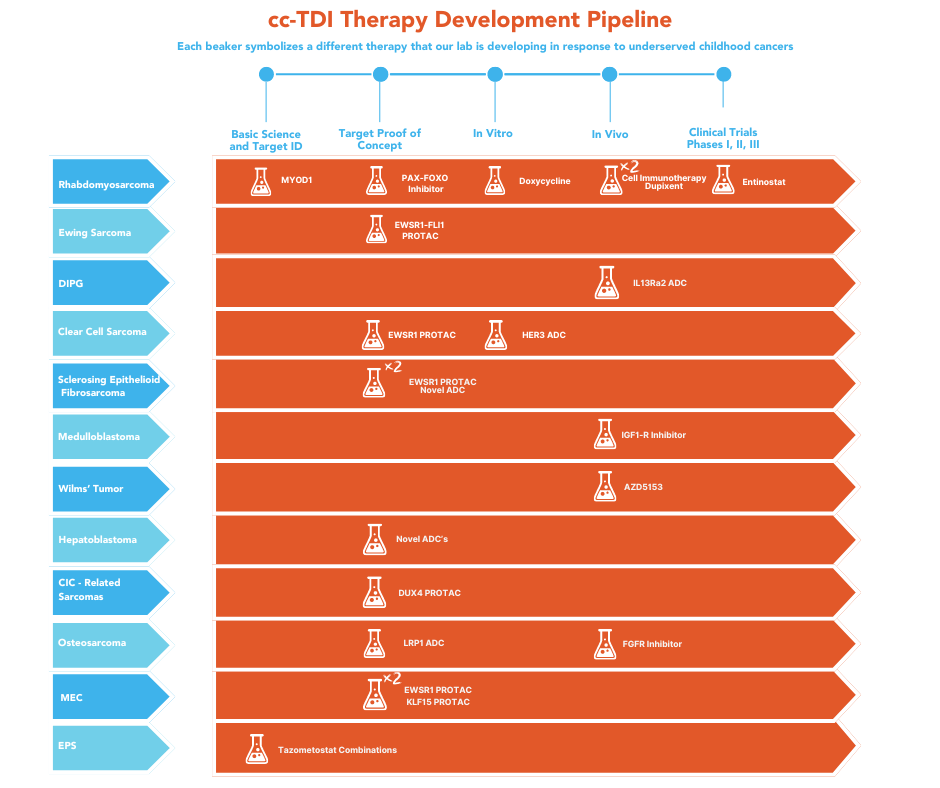

Timelines:
– For target proof of concept, 4-18 months
– For repurposed drugs, no extra time is required
– For in-house drugs being moved from a “hit” compound to a lead compound, add at least 24 months
– For in vitro proof of concept, 4-9 months
– For in vivo proof of concept, 12-18 months
– For Phase I, II or III clinical trials, the time to enroll patients and observe their responses for a curative-intent trial with definitive results can be 4-7 years.

